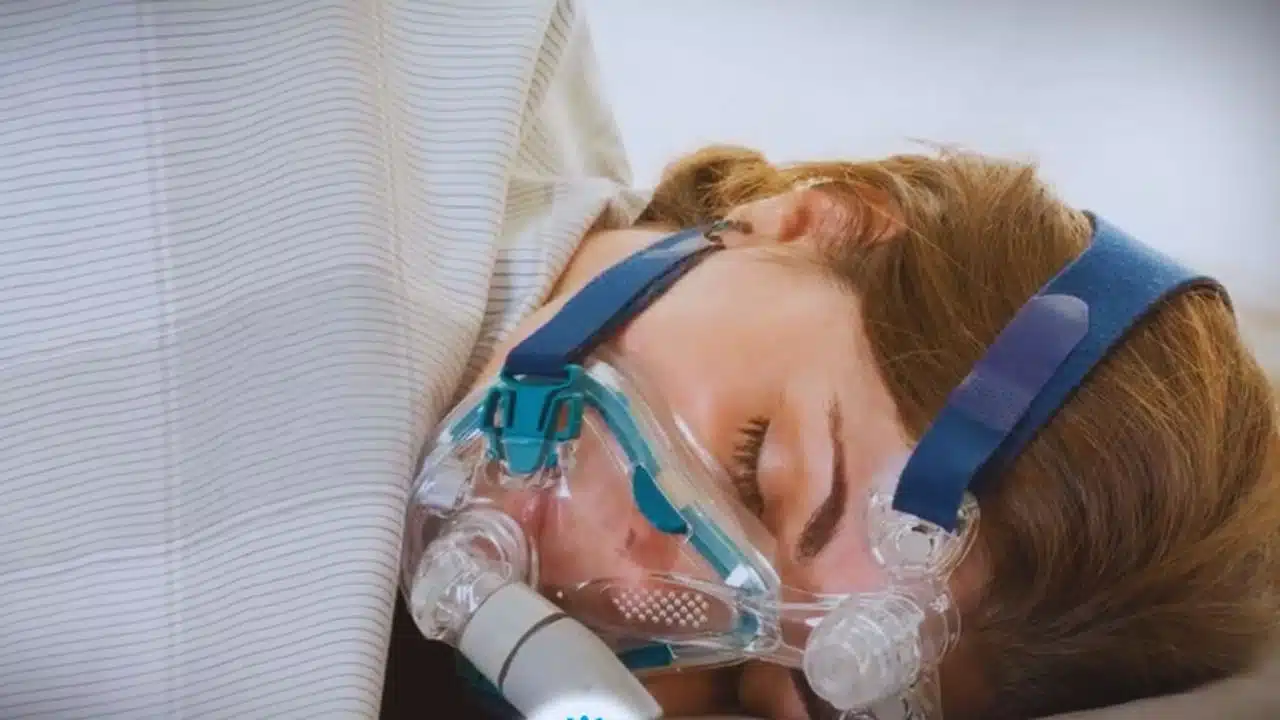
A popular weight loss drug, Tirzepatide, may offer a new avenue for treating sleep apnea, according to a recent study. The medication, which is found in the medicines Zepbound and Mounjaro, has shown promising results in reducing the severity of sleep apnea among patients with obesity.
Study Details and Findings
The study, published in the New England Journal of Medicine, involved two controlled trials with adults suffering from moderate to severe obstructive sleep apnea and obesity.
The research, funded by Eli Lilly and Co., the maker of Tirzepatide, found that the drug significantly reduced the apnea-hypopnea index (a measure of sleep apnea severity), body weight, and blood pressure and improved overall sleep quality.
Patients who took Tirzepatide experienced a reduction in sleep apnea episodes by about half to nearly 60%, compared to only a 10% reduction in those who received a placebo.
Additionally, participants on the drug lost between 18% and 20% of their body weight, which likely contributed to the improvements in sleep apnea symptoms.
FDA Approval and Expert Opinions
Eli Lilly and Co. has submitted a request to the Food and Drug Administration (FDA) to expand the use of Tirzepatide to include the treatment of moderate to severe sleep apnea.
A decision from the FDA is expected by the end of the year. However, experts caution that more research is needed to determine if Tirzepatide can be used as a sole treatment for sleep apnea.
The Impact of Sleep Apne
Sleep apnea affects an estimated 20 million Americans. The disorder, characterized by repeated interruptions in breathing during sleep, can lead to short-term issues such as snoring, brain fog, and daytime sleepiness.
More concerning, however, are the long-term risks, which include heart disease, dementia, and even death.
Clinical Trials and Results
The clinical trials, part of the SURMOUNT-OSA study, were multi-center, randomized, double-blind, and placebo-controlled, involving 469 patients.
These trials were conducted over a period of 52 weeks. In the first trial, patients with obstructive sleep apnea (OSA) and obesity who were not yet using positive airway pressure (PAP) therapy showed significantly reduced apnea events.
The baseline number of events was 51.5 per hour, which dropped to 36 per hour in the Tirzepatide group—a nearly 30% reduction. The placebo group saw less than a 10% reduction.
In the second trial, which included patients already using PAP therapy, the results were even more compelling.
The baseline number of events was 49.5 per hour, which dropped to 20.2 per hour in the Tirzepatide group after 52 weeks—a reduction of nearly two-thirds. Again, the placebo group saw a decrease of only about 10%.
Potential Market and Future Research
The study’s lead author, Atul Malhotra, highlighted the potential for Tirzepatide to transform the treatment approach for sleep apnea. If approved, Tirzepatide could become the first pharmaceutical treatment targeting the underlying causes of sleep apnea.
This is significant, as there are currently no FDA-approved pharmacological treatments for OSA, with CPAP therapy being the current gold standard.
Eli Lilly aims to begin submitting data to the FDA and other global regulators mid-year. With an estimated one billion people worldwide suffering from obstructive sleep apnea, the potential market for Tirzepatide is substantial, particularly as many cases go undiagnosed and untreated.
The findings from the study indicate that Tirzepatide could be a groundbreaking treatment for obstructive sleep apnea, offering significant improvements for patients with obesity.
While the results are promising, further research is needed to understand the drug’s potential fully and to confirm its efficacy as a standalone treatment. As the FDA reviews the data, the possibility of a new, effective treatment for sleep apnea brings hope to millions of sufferers.
The information is taken from The Hill and NBC News